Psittacine Pediatrics: Housing and Feeding of Baby Parrots
Introduction
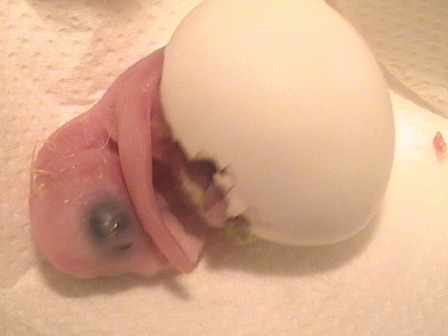 Hand-feeding baby birds is an vital part of any successful aviculture operation. Psittacine eggs that are artificially incubated or babies pulled from their parents at a young age, must be hand-fed for three to five months. Hand-fed birds make tamer companions and increase production if the parents re-clutch. Breeding pairs may neglect all or the youngest of their babies or they can cannibalise them. With these pairs there is no choice but to pull the babies and hand-fed.
Hand-feeding baby birds is an vital part of any successful aviculture operation. Psittacine eggs that are artificially incubated or babies pulled from their parents at a young age, must be hand-fed for three to five months. Hand-fed birds make tamer companions and increase production if the parents re-clutch. Breeding pairs may neglect all or the youngest of their babies or they can cannibalise them. With these pairs there is no choice but to pull the babies and hand-fed.
Most exotic birds kept in captivity, such as psittacines and the many types of “soft bills” are altricial that is their young are hatched blind, helpless in food gathering and are unable to thermoregulate. Since poultry are precocial, little information on their relatively easy care is directly useful for parrots. Aviculturists have by trial and error methods, developed procedures to successfully raise hatchlings right from hatching.
Hygiene and Baby Movement
Nursery management must have as the main goal disease prevention. With chicks lacking fully developed immune systems, normal gut flora just becoming established, and parent pairs previously exposed to many different organisms, stress due to poor feeding or inadequate environment can quickly lead to disease. The nursery usually has a concentration of such susceptible individuals so there is greater chance of an epornitic occurring here, crippling the cash flow of an operation.
The flow of babies within a nursery can reduce the chances of cross-contamination. There should be no mixing of parent raised babies, even if the parents appear perfectly healthy and have been with the breeder for many years. Subclinical carriers of Psittacine beak and feather disease, polyomavirus and other diseases may transmit these vertically (via the egg) but with greater probably horizontally (directly) to babies in the nest. If these babies are pulled and not properly handled within the nursery, significant mortality could result. Increasing the number of hatchlings from artificially incubated eggs can minimize the chance of parent birds infecting their offspring.
If parent fed chicks are to be hand-fed they should be pulled from the nest box before three weeks of age or the time of emergence of pin feathers on the wings. Older chicks will be stressed for the first few days in the strange nursery and may not want to be fed. However once they are hungry enough, usually about 12 hours later, they will accept hand-feeding.

Hands must be cleansed before and also between feeding and handling different babies and clutches. Disposable latex gloves can be changed between each group of babies or allow hands to be wiped with a disinfectant without irritating skin. Babies should not be removed from their containers unless necessary and a clean paper towel placed under each baby when weighing.
Food Preparation
A fresh batch of formula should be mixed up for each feeding. This is more hygienic and convenient since food does not need to be stored. To properly prepare the diet for feeding, a balance for weighing formula and accurate volumetric container for water should be used. Use accurate measuring devices, clean utensils and stir the food well.
Cooking time will vary slightly with model of microwave oven and type of container. Plastic containers seem to cool the food slower than ones made of glass. The final temperature of the food, just before feeding, should be slightly warmer than human body temperature or about 40°C (100°F). Be careful not to overheat the food as burning the babies can occur if the food contains hot spots or is only a few degrees too hot.
Table 1-
Water/Dry Mash Ratio of hand-feeding Formula
| Age of baby | Amount of dry mash | Volume of Water | |
| Hatching to 2 days | 1 tbs = 9 g (15 cc) volume rario 1:5 weight ratio 1:7 (12.5% Solids) |
72 ml |  |
| 3 days to weaning | 1/2 cup = 64 g (120 cc) volume ratio 1:2 weight ratio 1:4 (20% Solids) |
240 ml |  |
Formulas for young babies, up to day two to four, should be significantly more diluted, about 5 to 10 percent solids. These young babies may also develop better on less rich diets having lower fat and protein and more easily digested carbohydrates (another area requiring research). Older babies should receive formula with a solid content in the 20 to 30 per cent range which usually results in a consistency a little thinner than apple sauce. Do not assume that such a texture represents the correct nutrient density as thickeners can make a formula with low dry matter levels appear much denser. Formula can be maintained at the correct feeding temperature by setting it in a bowl of hot water.

Methods of Feeding
There are several possible techniques of feeding young parrots. Psittacine chicks produce a feeding response when the commissures of the beak are touched.  This bobbing action closes the glottis and food passes into the crop. A bent teaspoon can be used to feed the formula but this can be a messy and time consuming way of feeding. Spoon feeding may produce a tamer bird because of the increased handling and is the preferred method of feeding of some of the most experienced aviculturists in the world (Low, 1987). However, there may be a greater chance of disease transmission with spoon feeding as the spoon is repeatedly touching the baby’s mouth and then dipped into the formula thus contaminating the food for the other babies.
This bobbing action closes the glottis and food passes into the crop. A bent teaspoon can be used to feed the formula but this can be a messy and time consuming way of feeding. Spoon feeding may produce a tamer bird because of the increased handling and is the preferred method of feeding of some of the most experienced aviculturists in the world (Low, 1987). However, there may be a greater chance of disease transmission with spoon feeding as the spoon is repeatedly touching the baby’s mouth and then dipped into the formula thus contaminating the food for the other babies.
Small plastic pipettes are used by some breeders to feed younger babies. Since these pipettes can only hold a few ml of food they require many repeated dippings into the food container with the same potential for disease spread as with spoons.
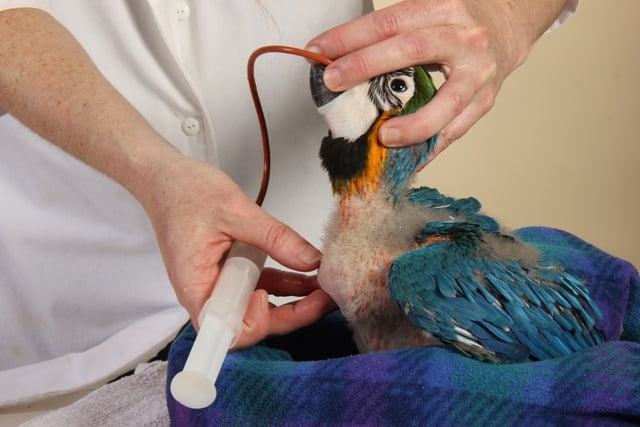
Catheter tipped syringes of various sizes are becoming popular instruments in feeding baby birds. Syringes can be used to slowly dribble the food into the mouth of the bird. Syringes allow the measuring of the amount of food fed and are easier to use. Silicone rubber and “O” ring syringes last longer than black rubber ones. Better still are syringes with no rubber gaskets and simply a concave round end. Soaking in tamed iodine disinfectant between feedings.
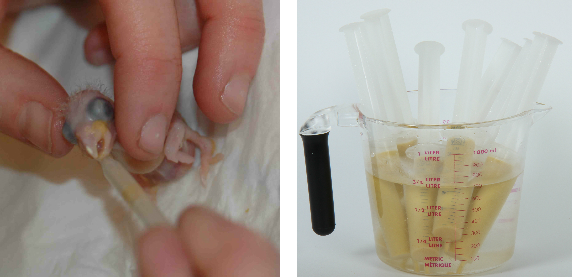
Syringes are usually used to shoot the food into the crop of the bird. Hold the baby’s head loosely by placing a finger on both sides of the beak and the back of the head. Place the tip of the feeding syringe into the left side of the birds mouth and once the bird gives a feeding response (head pumping) shoot the food towards the back of the head. This procedure is relatively safe but requires some practice before it is done without a mess. The advantage of syringes is that a separate one can be used for each clutch of babies so that if one pair produces diseased offspring it doesn’t spread to the other babies.
A short soft rubber tube can be attached to the tip of the syringe which can then be placed further back into the mouth to prevent food going down the trachea. However this may have a behavioral effect on the baby and may prolong the weaning process since this is the least natural of all the feeding methods.

Fill the crop well without over stretching it and allow it to completely empty between feedings. Do not confuse the small pouch of loose skin around an empty crop as a crop that still contains food. Babies have looser skin around the crop to allow it to stretch out. Sick babies are more likely to aspirate themselves and the crop becomes flaccid. These babies should be placed in small containers, like disposable plastic cups, which hold them up prevent them from falling on their crop. Feeding frequency or gut transit time is dependent on the percent solids of the formula, its digestibility and caloric density. Babies less than 3 days old get fed six times a day and older ones 4 times a day.
Metal feeding tubes are only necessary when force feeding sick birds.
Whatever implements are used they should be disinfected between feedings and replaced periodically. Some aviculturists boil syringes in large pressure cookers or in regular pots but more commonly cold disinfectant baths are used to soak feeding equipment. Gluteraldehyde based products appear the strongest disinfectants however quartenary ammonium compounds or iodophors are acceptable under normal conditions. Phenol based disinfectants are very irritating to skin and chlorhexidines do not effectively kill pseudomonas bacteria which are common in wet environments and water. At HARI we lost several two month old cockatoos to pseudomonas when we used chlorhexidine to disinfect syringes. The bacteria were cultured directly from the syringes and upon close examination colonies could be seen in the corners of the syringe shafts.
HOUSING BABIES
Brooders and Containers
There are as many different and successful baby brooders as there are hand-feeding formulas. Some are homemade wooden boxes with electric elements or lights to keep the babies warm. Others are adapted metal game chick brooders or glass aquariums with custom made heaters to partially cover the top or bottom of the tank. Brooders set up with heating pads often result in cooked babies, thermal injuries or cold babies since precise, constant temperature maintenance is difficult (Stoddard, 1988). Another problem with homemade or adapted brooders is their usual lack of humidity control. Human baby incubators and specific commercial baby bird brooders have more accurate temperature and humidity control but are difficult to obtain or can become expensive when many babies have to be housed.
 Brooders should be relatively small as an important part of disease prevention with babies is to keep clutches separate and this is limited with large brooders. They should provide constant, even heat that can be finely adjusted, be well ventilated, easily cleaned and have a water receptacle to add humidity.
Brooders should be relatively small as an important part of disease prevention with babies is to keep clutches separate and this is limited with large brooders. They should provide constant, even heat that can be finely adjusted, be well ventilated, easily cleaned and have a water receptacle to add humidity.
One set-up which meets many of these requirements uses small aquariums to contain babies which is then placed a much larger aquarium or plastic pan filled with about three or four inches of heated water. The water is maintained at 40°C (100°F) with a submersible thermostat aquarium heater. Salt should be added to the water to keep down the growth of pseudomonas and other pathogenic bacteria. Evaporated water must be replaced so that the horizontally laid heater remains submerged.
Bedding
Young babies, less than two weeks of age, can be kept on paper towels in plastic cups, re-used yogurt containers or small aquariums. Babies should be sitting on clean and dry bedding which is therefore changed at each feeding. This may not provide firm enough footing for some birds which should be transferred onto paper or wood shavings or towels to avoid splayed legs. When babies are two-three weeks old, we transfer the babies to a small plastic containers that contain a few layers of newspaper under a few inches of wood shavings.Corn cob, walnut shell and pellet type bedding are not popular or losing favour with aviculturists because of various drawbacks.
Some babies may have a tendency to eat shavings, perhaps as a result of other complications such as calorie deficient diets or excess heat. Using towels has the advantage of being able to clearly see the baby’s droppings but the feet and feathers can get caked up with feces and time and energy must be spent on washing towels. Disposable diapers are expensive and wasteful. Babies stay cleaner when they are kept on processed paper product or shavings.
Temperature
The ambient temperature and humidity of baby altricial birds must be regulated. Young, up until they are feathered out, need supplemental heat above room temperature to thrive and even survive. Chilled babies, either because of neglectful parent birds or power failure to the brooder heater, will deteriorate quickly. These chicks may die later even after being warmed up. Nursery room temperature should be kept warm between 78 and 82 degrees fahrenheit.
The brooder temperature for recently hatched chicks can remain at the hatching temperature of 35.0°-36.5°C (96°-98°F) for the first few days. Once the baby is eating more solid food, at about 2-3 days of age, it should be kept at a lower temperature of 33.5°-35.0°C (92°-96°F) depending on the species and its metabolism. From there up to about two weeks of age babies should be in an environmental temperature of 32.0°-33.5°C (90°-92°F) (Table 2). If temperatures are to high the chick may exhibit panting, unrest, hyperactivity and have dry, reddened skin (Clubb and Clubb, 1986). Cold temperatures may result in death, poor gut motility, crop stasis or other digestive disorders, failure to feed or beg, inactivity or shivering (Clubb and Clubb, 1986).
Table 2-
Approximate Brooder Temperature
| Age | Temperature °C | Temperature °F |
| Hatch to Day 2-3 | 35.0-36.5 | 96-98 |
| Day 3 to Day 14-21 | 31.1-34.0 | 88-94 |
| 3 weeks to Weaning | 25.0-30.0 | 76-86 |
Humidity
Natural nesting cavities in wooden tree trunks probably have a high relative humidity. Moist droppings from any babies within them will add to this, resulting in an environment of high humidity. These are unfortunately not the humidity levels found in most nursery brooders (Clipsham, 1989b). Most babies are traditionally raised on dry heat with heating pads, light bulbs or electric coils.
A range of 55-70% humidity produces quieter, fatter babies with a greater growth rate than those kept at levels of 15-35% (Clipsham, 1989b). Ambient humidity for hatching eggs and hand fed chicks can be increased by increasing nursery room humidity and, more effectively, by using containers of water as a source of both heat and humidity. A small aquarium submerged in and surrounded by a water bath heated by a submersible aquarium heater is very effective for eggs and up to one week old babies.
Identifying the Chicks
Babies must be properly identified in order to follow the linage and pair unrelated birds in the future. If considerable numbers of birds are being raised it becomes difficult to maintain the identity of similar babies from different parents. Several companies make closed leg bands in many different sizes. These bands can only be slipped to the birds leg when their feet are a small size. The best time to apply the bands is at about 2 – 3 weeks of age. Place the three larger toes through the appropriate sized band and then slide the band over the small inside toe which is held back against the foot.
Microchips implants are now available but are not yet commonly used for identifying baby birds.
Record Keeping and Growth Rates
The goal of any exotic aviculture venture is to raise healthy babies with minimum losses. Thus achieving the fastest growth rates possible on a particular formula are not as critical as the proper development of the baby. Average weight gains for the particular diet being used do help to monitor the development of individual babies. Weights taken each morning before the first feeding are less influenced by residual food still in the gut of the bird from the previous feeding as babies are usually allowed to empty over night. The time between feedings should slowly lengthen as the baby gets older but any sudden slow down in digestion is an indication of illness.
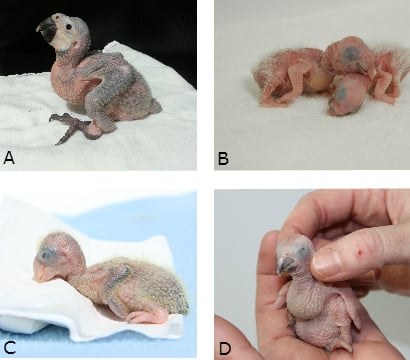
Stoddard (1988) monitors the health and development of young babies by the;
- Plumpness of the toes, wings and rump.
- Skin colour – should be a flesh-toned pink.
- Skin texture – should be translucent and soft.
- Anatomical symmetry – malnourished babies often have thin feet, toes, and wings as well as a disproportionately large head.
Flammmer (1986) lists other physical characteristics of nestling psittacines.
Complications
1 – Crop Stasis
The crop should completely empty between feedings. Food that remains in the crop too long may “sour” and provide an excellent growth media for opportunist bacteria and fungi.
Hard lumps may form in the crops of some babies if the solid matter of the hand feeding formula separates from water. Treatment consists of feeding a little warm water and massaging the crop/lump until it is dissolved. The next feeding should consist of diluted formula following which the crop should be back to normal.
Under feeding may result in result in some babies ingesting bedding material. In a case of eclectus parrots ingesting wood-shavings they all died due to the gizzard damming up, preventing normal digestion (Smith, 1985).
2 – Crop Burn
If the temperature of the food is greater than 41°C (105°F) it may scald the crop and cause necrosis and fistulation (Giddings, 1986). Even with careful mixing and cooking some of the most experienced facilities may burn babies’ crops by feeding hot formula. Microwaves are usually used when overcooked food is fed. Hot areas within the food may go undetected even with a thermometer. It is best to let the formula stand for a minute, mix well and double check the temperature.
3 – Constricted Toes
Avascular digital necrosis or “big toe” is seen in baby macaws, eclectus and african greys. A ring of fibrous tissue may be initiated by rapid loss of body fluids from a crack of skin and encircles the toe leading to a constriction (Clipsham, 1989b).
Higher ambient humidity levels are reported to decrease this problem (Clipsham, 1989b; Joyner, 1987).
4 – Aspiration
On several occasions very young, less than 3 days old, weak babies were accidentally aspirated at HARI. This may have been due to poor feeding responses and force feeding. It is also possible to drop a baby onto its full crop resulting in the food being forced out and into the buccal cavity. The baby, not expecting food at that time may aspirate it. Feeding older birds by a tube placed into the crop will decrease the likelihood of aspiration in the more difficult species to feed (Joyner, 1987), such as 2 month old cockatoos.
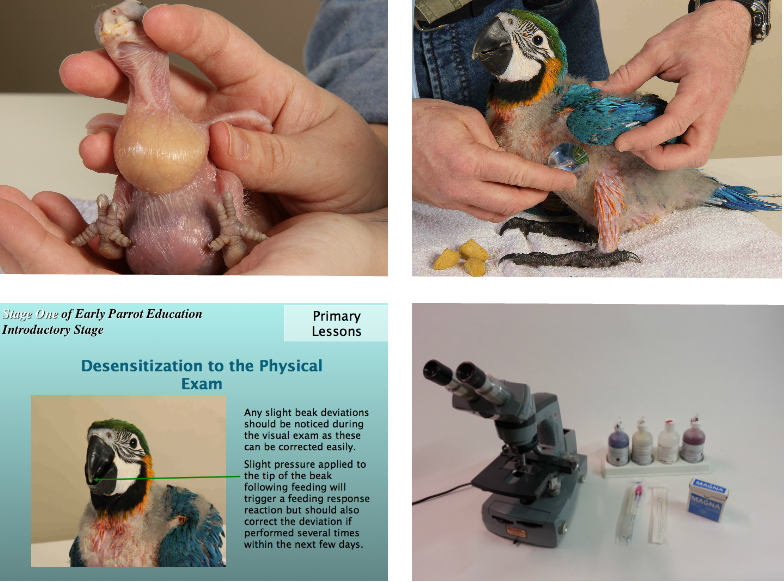
5 – Beak Deformities
At HARI a lateral beak deformity has occurred in a baby macaw and an ingrown upper beak in a cockatoo. These types of deformities have been observed with other aviculturists (Joyner, 1987; Clubb and Clubb, 1989). The etiology of these deviations is not known but may be related to unnatural feeding methods or poor nutrition. The rapid growth and strong feeding response of macaws may exacerbate any deviations in growth.
6 – Bacterial, Fungal and Viral Diseases
Gram negative bacteria such as E. coli, Klebsiella sp., and Pseudomonas sp., are commonly considered pathogens and are often associated with disease (Clubb and Clubb, 1986). The “normal” aerobic alimentary tract flora for baby psittacines may include Lactobacillus sp., Staphylococcus eipdermidis, Streptococcus sp., Corynebactrium sp. and Bacillus sp. (Drewes, 1983). Candidiasis, caused by Candida albicans, is a yeast infection, usually of the crop. It is often a secondary problem associated with slowing of gut transit time. Digestive function may be upset by bacterial infections or blockage of the crop opening with foreign matter such as bedding substrate. Food which remains in the crop too long may begin to ferment leading to a “sour crop”. Dietary causes of a slow down in the digestive tract include food too cold, food with inadequate moisture content or ability to hold water (gelatin quality), food too high in fat or protein or too low in fiber. Babies will often vomit if one of these is not quite right.
Abnormally high Candida infections in Palm Cockatoos may have been due to the simple sugars in the sweetened applesauce and sugar ingredients of the diet used by the New York Zoological Society (Sheppard and Turner, 1987).
Viral diseases can be prevented by only handfeeding incubator hatched babies and leaving parentally hatched nestlings with their parents. A separate nursery for babies exposed to parental feedings could be another alternative.
Weaning
Weaning appears not to be a learned process and occurs at a certain age which is not affected by external stimulation of hunger (Roudybush, 1986). Cockatiel chicks which reach a higher maximum weight sooner also wean earlier than chicks which do not gain more than their normal adult weight (Roudybush, 1986). Babies may lose weight (up to 10-15%) during weaning although keeping weight on them is probably better for birds to be shipped out unweaned.
When the babies have about an inch of blood left in their wings they are transferred to a cage which has small openings on the bottom wire for the babies to walk on comfortably. Perches should initially be near the bottom of the cage for the uncoordinated fledglings.
 Once babies have absorbed the blood in their wing feathers they are ready to be placed into a cage. The cage should be large enough to allow the birds to exercise their wings. Perches are better placed near the bottom of the cage where young birds first spend most of their time. A cup of formulated parrot food, moistened with hot water, appears to be easier for babies to wean onto than seeds or hard vegetables. This cup must be made fresh daily (or more often in hot climates) to avoid spoilage and should be placed near where they perch.
Once babies have absorbed the blood in their wing feathers they are ready to be placed into a cage. The cage should be large enough to allow the birds to exercise their wings. Perches are better placed near the bottom of the cage where young birds first spend most of their time. A cup of formulated parrot food, moistened with hot water, appears to be easier for babies to wean onto than seeds or hard vegetables. This cup must be made fresh daily (or more often in hot climates) to avoid spoilage and should be placed near where they perch.
To assist in weaning, feed a little amount of formula with a spoon from the cup that the birds should begin to eat from. Additional syringes of food should be given two or three times a day so they do not lose do much weight but avoid always feeding when babies beg or this behaviour may increase.
Potential for Breeding
There is concern that hand-rearing imprints the birds on humans and they will therefore not be suitable for breeding when they become sexually mature. Third generation breeding of Amazona leucocephala by Ramon Noegel indicates that this is not the case. Hand-reared birds often mature and breed in a shorter period than it takes wild-caught adults to settle down. Captive raised birds lack the stress that causes wild birds not to breed or abandon their nests.
 If the babies birds are to be kept for breeding it is probably better to raise them in groups rather than individually.
If the babies birds are to be kept for breeding it is probably better to raise them in groups rather than individually.
Research with cockatiels has shown that early rearing experience is important for males to learn characteristics of the opposite sex, and for males and females to learn characteristics of nest-sites (Myers et al., 1988).
Care of Baby Parrot
Environment
A newly arrived baby parrot should be kept in a warm and quiet place for the first few weeks. A young bird needs to rest during the day and a busy environment may exhaust it. This stress weakens the immune system which may result in disease(s).

Provide your bird with a cage big enough so it can fully expand its wings and perhaps jump between perches. Parrots like to climb around their cage and like its security when they rest. Place several toys in its cage, to avoid boredom and change these regularly.
When outside of its cage, you can put your bird on a T perch but this should not be its only territory, since it cannot move around much.
Feeding
A sudden change of diet could lead to weight loss and digestive upset with subsequent problems, so, it is best to continue with the same diet.
If the baby bird is not weaned, follow the directions on the leaflet of the Tropican hand-feeding formula and provide three food bowls to the baby: one filled with dry Tropican granules, one with soft Tropican moisten with warm water, and a bowl of fresh water. Moist food spoils very fast; so be sure to clean the bowl twice a day. Always use the same bowls for your baby birds unless bowls used by another bird has been thoroughly disinfected.
Exerci se
se
The bird should be allowed out of its cage to interact with people and to exercise each day. Play with the baby every day but do not spoil it too much initially or it will demand this attention later. Do not leave doors or windows open or go outside with an unclipped bird. You can clip the wing feathers but when doing this for the first time, ask someone who is familiar with clipping.
Medical Care
Captive bred birds are not used to the microorganisms carried by a wild caught exotic bird and you should avoid the contact between the two. Species susceptible to Pacheco and Pox diseases should be vaccinated against them if they will be in places with other birds at proximity such as in a pet shop, veterinary clinic, boarding facility or bird shows.
If your bird shows one or several of the following symptoms, it may be sick: sleeping during its usual peak activity, not eating or eating less than usual, diarrhea and feathers puffed up. In that situation, contact immediately your usual avian veterinarian. Isolate the bird. Keep it warm (30°C – 86°F) and try to give it some food.
References
BUCHER, T.L. (1983). Parrot eggs, embryos, and nestling: patterns and energetics growth and development. Physiol. Zool. 56 465-483.
CACCAMISE, D.F. (1975). Growth rate in the Monk Parakeet. The Wilson Bulletin 88 495-497.
CLIPSHAM, R. (1989a). Pediatric management and medicine. Avian Veterinarians. 1:10-13.
CLIPSHAM, R. (1989b). Preventive Aviary Medical Management. Proceedings of the American Federation of Aviculture Veterinary Seminar, pp 15-28.
CLUBB, S. L., and CLUBB, K. J. (1986). Psittacine pediatrics. Proceedings of the Association of Avian Veterinarians pp 317-332.
CLUBB, S.L. and CLUBB, K.J. (1989). Selected problems in psittacine pediatrics. Proceedings of the American Federation of Aviculture Veterinary Seminar, pp 29-35.
DREWES, L., and FLAMMER, K. (1983). Preliminary data on aerobic microflora of baby psittacine birds. Proceedings of the Jean Delacour/ IFCB Symposium on Breeding Birds in Captivity, pp 73-81.
FLAMMER, K. (1986). Pediatric medicine. In: Clinical Avian Medicine and Surgery: including aviculture. Eds. G. J. Harrison and L. R. Harrison, W. B. Saunders Company, Philadelphia, pp 634-650.
GIDDINGS, R. F. (1986). An avoidable cause of crop necrosis in nestling cockatiels. Veterinary Medicine 81:1025-1026.
JOYNER, K.L. (1987). Avicultural Pediatrics. American Federation of Aviculture Veterinary Seminar pp 22-33.
JOYNER, K.L. (1988). The use of a lactobacillus product in a psittacine hand-feeding diet; its effect on normal aerobic microflora early weight gain, and health. Proceedings Association of Avian Veterinarians pp 127-137.
LOW, R. (1987). Hand-Rearing Parrots. Blandford Press. Dorset, U.K.
MOSTERT, PETER (1989). Personal communication.
MYERS, S.A., MILLAM, J.R., ROUDYBUSH, T.E. and GRAU, C.R. (1988). Reproductive success of hand-reared vs. parent-reared cockatiels (Nymphicus Hollandicus). The Auk 105:536-542.
SHEPPARD, C. and TURNER, W. (1987). Handrearing Palm Cockatoos. AAZPA Annual Proceedings pp. 270-278.
SILVA, T. (1989). A monograph of endangered parrots. Hand-rearing. Silvio Mattacchione and Co., Pickering, Ontario, pp 23-26.
SMITH, G. A. (1985). Problems encountered in hand-rearing parrots. In: Cage and Aviary Bird Medicine Seminar, Australian Veterinary Poultry Association. pp 71-77.
SMITH, R.E. (1986). Psittacine beak and feather disease: a cluster of cases in a cockatoo breeding facility. Proceedings Association of Avian Veterinarians pp 17-20.
STODDARD, H.L. (1988). Avian Pediatric Seminar. Avian Pediatric Seminar Proceedings (Supplement) pp 1-19.
TAKESHITA, D.L., GRAHAM, L. and SILVERMAN, S. (1986). Hypervitaminosis D in baby macaws. Proceedings Association of Avian Veterinarians pp 341-346.
THOMPSON, D.R. AND BARBER, L. (1983). Successful techniques for hand raising young psittacine birds. Proceedings of the Jean Delacour/ IFCB Symposium on Breeding Birds in Captivity, pp 171-178.
By Mark Hagen, M.Ag.
Director of Research